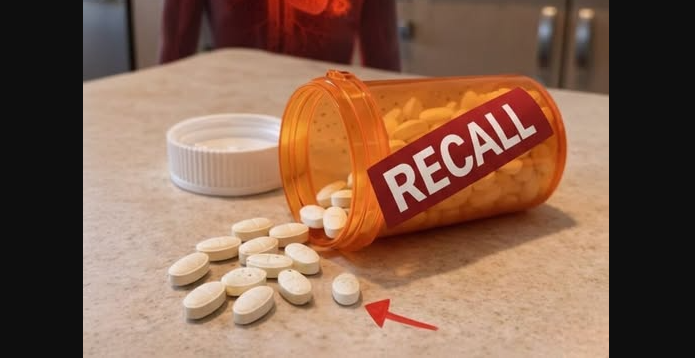
A recent recall of more than 600,000 bottles of the blood pressure medication Ramipril across the United States has raised awareness about how medications are produced and monitored. Federal regulators issued the recall after identifying concerns related to ingredient sourcing during manufacturing.
What Led to the Recall?
The affected products were manufactured by Lupin Pharmaceuticals. According to officials, some ingredients used in production came from a facility in India that had not undergone proper inspection or approval. Because quality standards could not be fully verified, regulators chose to remove the impacted batches as a precaution.
Importantly, the recall is not linked to confirmed injuries or health issues. Instead, it reflects a proactive effort to maintain strict safety standards in pharmaceutical production.
Understanding Ramipril
Ramipril is part of a group of medications known as ACE inhibitors. These drugs are widely prescribed to help manage high blood pressure and support overall heart health. By relaxing blood vessels, ramipril helps improve blood flow and reduce strain on the cardiovascular system.
Millions of patients rely on this medication to lower the risk of serious conditions such as heart attacks, strokes, and complications related to kidney disease.
Scope of the Recall
The recall includes ramipril capsules in three common strengths: 2.5 mg, 5 mg, and 10 mg. The affected bottles contain 90, 100, or 500 capsules, with expiration dates extending through July 2026. These products were distributed broadly to pharmacies and healthcare providers across the country.
What Patients Should Do
Health professionals recommend staying calm and informed. If you take ramipril, check your medication label for manufacturer details, strength, and expiration date, and speak with your pharmacist to confirm whether your medication is affected. If you believe your prescription is part of the recall, contacting your healthcare provider is the next step.
It’s important not to stop taking blood pressure medication without medical advice, as this can lead to sudden increases in blood pressure and potential health risks.
Alternative Treatment Options
If a replacement is needed, doctors may recommend other ACE inhibitors such as Lisinopril or Enalapril. These medications work in similar ways and are commonly used as alternatives, with your healthcare provider determining the most appropriate option.
Why Manufacturing Oversight Matters
This recall highlights the complexity of the global pharmaceutical supply chain. Many medications rely on ingredients sourced from multiple countries, making regulatory oversight essential.
Agencies conduct inspections to ensure facilities meet strict safety and quality standards. When a facility has not been properly evaluated, uncertainty about product quality can arise, prompting precautionary recalls like this one.
Staying Safe and Informed
While medication recalls can be concerning, they are an important part of protecting public health. Quick action helps reduce potential risks and maintain trust in the healthcare system.
Patients are encouraged to stay informed through reliable sources and continue following their prescribed treatment plans. Pharmacists and healthcare providers remain the best points of contact for any concerns.
With proper guidance, most patients can transition smoothly to alternative treatments if needed, ensuring that blood pressure management remains safe and effective.